Media
Company news


Team ChemDiv, Inc. at AACR 2024, San Diego!
Last week, our team had the incredible opportunity to attend the Annual Meeting of the American Association for Cancer Research (AACR) in San Diego.
Pop-Science
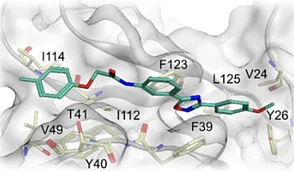

Novel Potent TIM-3 Inhibitors were discovered in ChemDiv’s collection.
The ChemDiv’s proprietary library of chemically diverse small molecule compounds was demonstrated to be a source of selective inhibitors targeting specific check points was found out in a recen...
ChemDiv in Publications


ChemDiv Company in publications and patents
We are passionate about the latest trends in life sciences and medicinal chemistry and closely monitoring the latest developments and discoveries in this area.
It means a lot to us if our custome...
Pharma News


Glenmark announces recall of high blood pressure medication in US
Glenmark Pharmaceuticals’ US-based unit has announced the recall of 6,528 bottles of Diltiazem Hydrochloride extended-release capsules for treating high blood pressure.
Follow us:


