Serine Proteases Inhibitors Library
Serine Protease Inhibitor Library
ChemDiv’s Serine protease inhibitor library is an extensive collection comprising 37,000 compounds.
Serine proteases are found in all living things and have a nucleophilic serine residue in their active site that attacks the carbonyl group of the substrate. This serine residue is the origin of their name. These enzymes are classified as endoproteases, known for catalyzing the hydrolysis of polypeptide bonds within the middle of peptide chains.
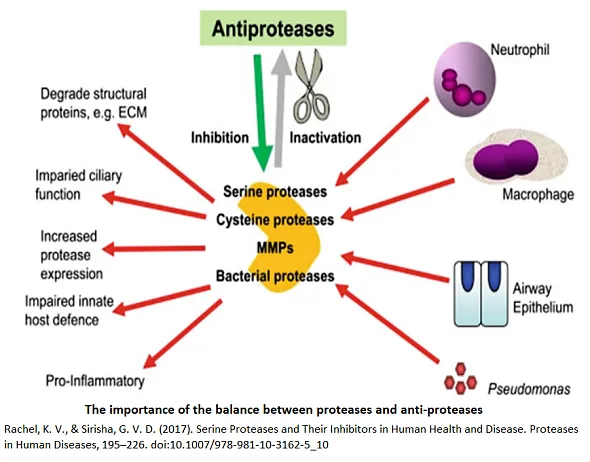
In the human immune system, cells such as endosomal vesicles express various serine proteases, including chymase and tryptase in mast cells, proteases in granulocytes, and granzymes in lymphocytes. Serine proteases derived from endosomal vesicles play pivotal roles in inflammation, tissue remodeling, apoptosis, and phagocytosis. Elevated activity of these proteases is linked to several pathological conditions, including allergies, autoimmune disorders, and cancer proliferation.
Serine protease inhibitors present an emerging area in drug discovery, targeting a broad spectrum of diseases characterized by abnormal protease activity. Their development, driven by a comprehensive understanding of protease structure and function, focuses on key therapeutic areas such as cancer, coagulation disorders, inflammatory and autoimmune diseases, cardiovascular diseases, and muscular dystrophy [1].
References:
[1] Rachel, K. V., & Sirisha, G. V. D. (2017). Serine Proteases and Their Inhibitors in Human Health and Disease. Proteases in Human Diseases, 195–226. doi:10.1007/978-981-10-3162-5_10