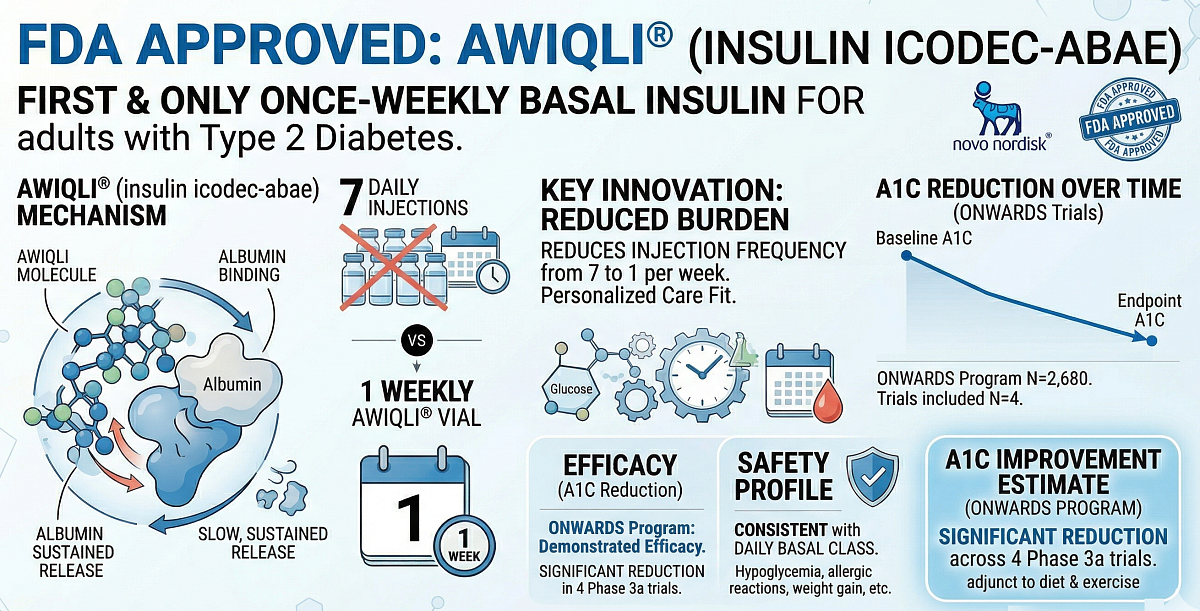
PLAINSBORO, N.J. -- Novo Nordisk today announced that the US Food and Drug Administration (FDA) has approved Awiqli® (insulin icodec-abae) injection 700 units/mL, the first and only once-weekly, long-acting basal insulin, indicated as an adjunct to diet and exercise to improve glycemic control (blood sugar) in adults living with type 2 diabetes.
This new approval offers the only once‑weekly basal insulin option, recognizing the importance of personalized care and the need for treatments that fit different patient routines and preferences for adults living with type 2 diabetes.
"Awiqli® is an important new option that meets a real need as the first FDA-approved, once-weekly basal insulin for adult patients with type 2 diabetes. It's helping to reframe what basal insulin care can look like," said Anna Windle, PhD, Group Vice President, Clinical Development, Medical & Regulatory Affairs at Novo Nordisk Inc. "Awiqli® may address challenges associated with the frequency of daily basal injections, by reducing them from seven to one per week. It is an important advancement for adults with type 2 diabetes who may benefit from an alternative treatment option."
The ONWARDS Phase 3a Clinical Program
This approval is based on results from the ONWARDS type 2 diabetes phase 3a program for once-weekly Awiqli® injection which comprises four randomized, active-controlled, treat-to-target trials in approximately 2,680 adults with uncontrolled type 2 diabetes. In these trials, Awiqli® was used in combination with a mealtime insulin or in combination with common oral anti-diabetic agents and/or GLP-1 receptor agonists.
The clinical program evaluated once-weekly Awiqli® vs. daily basal insulin and demonstrated efficacy in the primary endpoint of reduction of A1C across the ONWARDS pivotal clinical trial program for adults with type 2 diabetes.
Safety Profile and Adverse Events
Across these ONWARDS trials, the safety profile of Awiqli® was overall consistent with the daily basal insulin class. Common adverse events include hypoglycemia (low blood sugar), serious allergic reactions (whole body reactions), reactions at the injection site, skin thickening or pits at the injection site (lipodystrophy), itching, rash, swelling of your hands and feet, and weight gain.