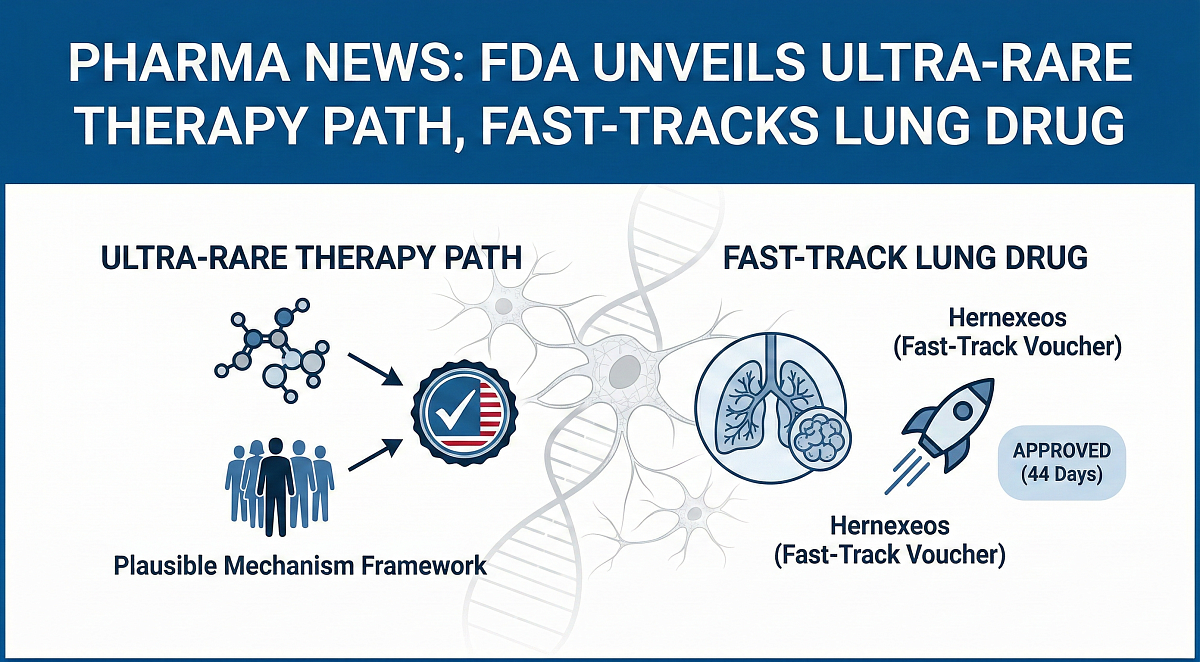
WASHINGTON, D.C. — The Food and Drug Administration (FDA) has released draft guidance outlining a new pathway to approve highly individualized treatments for ultra-rare diseases and has granted expedited approval to a lung cancer drug under a fast-track voucher program.
Drug developers may rely on a new “Plausible Mechanism Framework” when traditional large clinical trials are not feasible. The FDA approved the lung cancer drug Hernexeos 44 days after its application was filed. Public comments on the draft ultra-rare disease guidance are open for 60 days after publication in the Federal Register.
Addressing the Challenge of Small Patient Populations
The FDA on February 23 issued draft guidance describing how sponsors can generate substantial evidence of safety and effectiveness for targeted individualized therapies when randomized controlled trials are not practical due to extremely small patient populations.
Randomized controlled trials typically compare a new treatment to a placebo or standard therapy in large groups of patients. In ultra-rare diseases, the number of patients may be too small to conduct such trials.
The draft guidance, issued by the Center for Biologics Evaluation and Research and the Center for Drug Evaluation and Research, focuses on genome editing and RNA-based therapies such as antisense oligonucleotides — short strands of synthetic genetic material designed to alter how genes are expressed. The agency said the framework could apply to other tailored therapies that directly address a disease’s known biological cause.
The Plausible Mechanism Framework
The guidance introduces what FDA officials describe as a “Plausible Mechanism Framework,” under which approval could be supported by strong scientific evidence that a therapy corrects or modifies the underlying genetic, cellular or molecular abnormality causing the disease.
Key Elements Outlined in the Draft:
- Identifying the disease-causing abnormality.
- Demonstrating the therapy targets the root cause or biological pathway.
- Relying on well-characterized data from untreated patients.
- Confirming successful gene editing or molecular targeting.
Note: For traditional approval, the therapy should show improvement in clinical outcomes, disease progression, or biomarkers that are known to predict benefit.
The draft guidance, titled Considerations for the Use of the Plausible Mechanism Framework to Develop Individualized Therapies that Target Specific Genetic Conditions with Known Biological Cause, is available for public comment at Regulations.gov.