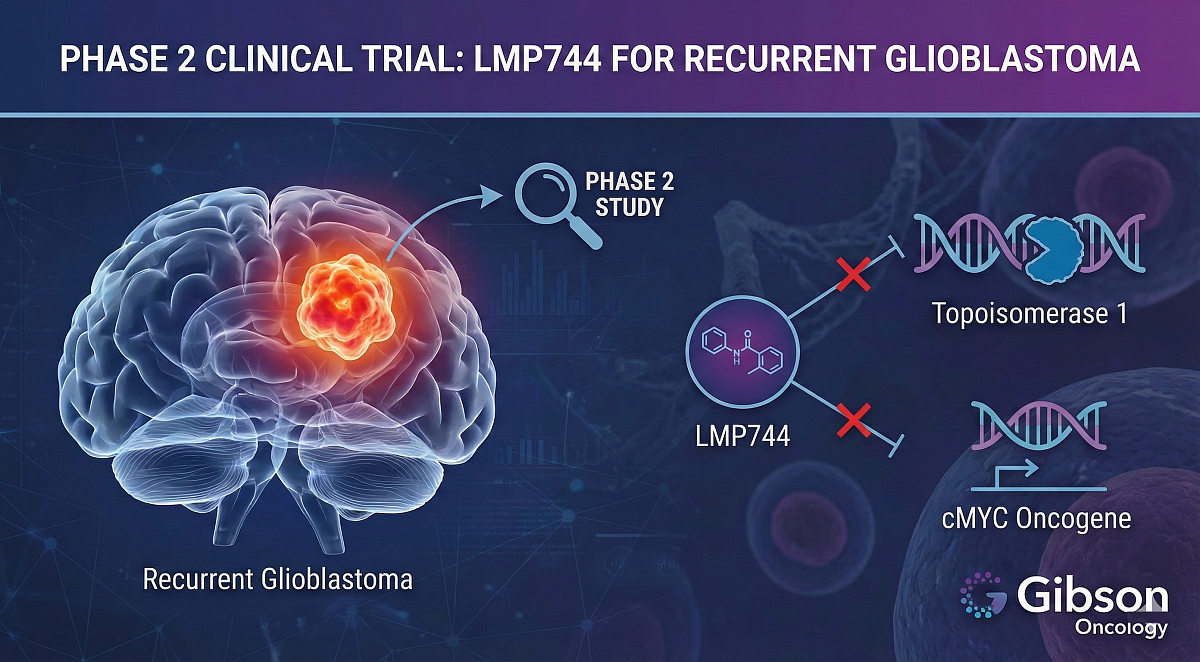
Gibson Oncology, a clinical-stage private pharmaceutical company headquartered in Miami, announced it has entered Phase 2 clinical trials with LMP744 (Synonyms: MJ-III65; NSC706744) for the treatment of first-time recurrent glioblastoma patients.
The company is focused on advancing its novel small molecules with dual-action inhibition of topoisomerase 1 and reduction of overexpression of the cMYC oncogene. The primary endpoint of the study is to evaluate tumor regression in patients with recurrent glioblastoma. Secondary endpoints will evaluate parameters such as progression-free survival, biological changes in glioblastoma tissues obtained pre- and post-treatment, self-reported quality of life, and overall survival.
Phase 2 Trial Design
During the Phase 2 trial, approximately 40 first-time recurrent glioblastoma patients will receive a once-a-day, one-hour infusion of LMP744 for five consecutive days, with biological analyses conducted on brain tissues obtained before and after this initial treatment period. If biological results are favorable, treatment will continue for 12 cycles, with each cycle consisting of five consecutive days of treatment followed by 23 days of no treatment.
Understanding Glioblastoma and Previous Clinical Efficacy
Glioblastomas are malignant tumors that develop in the brain or spinal cord. Tumors grow rapidly and invade nearby tissue. Standard treatment includes surgery, radiation, and chemotherapy. Life expectancy after diagnosis has been approximately 15-18 months for the past three decades.
Phase 1 Results: In two Phase 1 human trials involving over 40 heavily pretreated, advanced-stage cancer patients, LMP744 proved to be well tolerated and resulted in two patients whose tumors decreased at least 30% in size and 35% of patients whose tumors did not increase in size for a period of time lasting up to 18 months.
Gibson Oncology has extensive intellectual property and orphan drug designations on both LMP744 and LMP400, as well as pediatric designation on LMP400. LMP744 and LMP400 were designed and synthesized by a research group at Purdue University led by Mark Cushman, who serves as Gibson's chief scientific officer, founder, and board member.