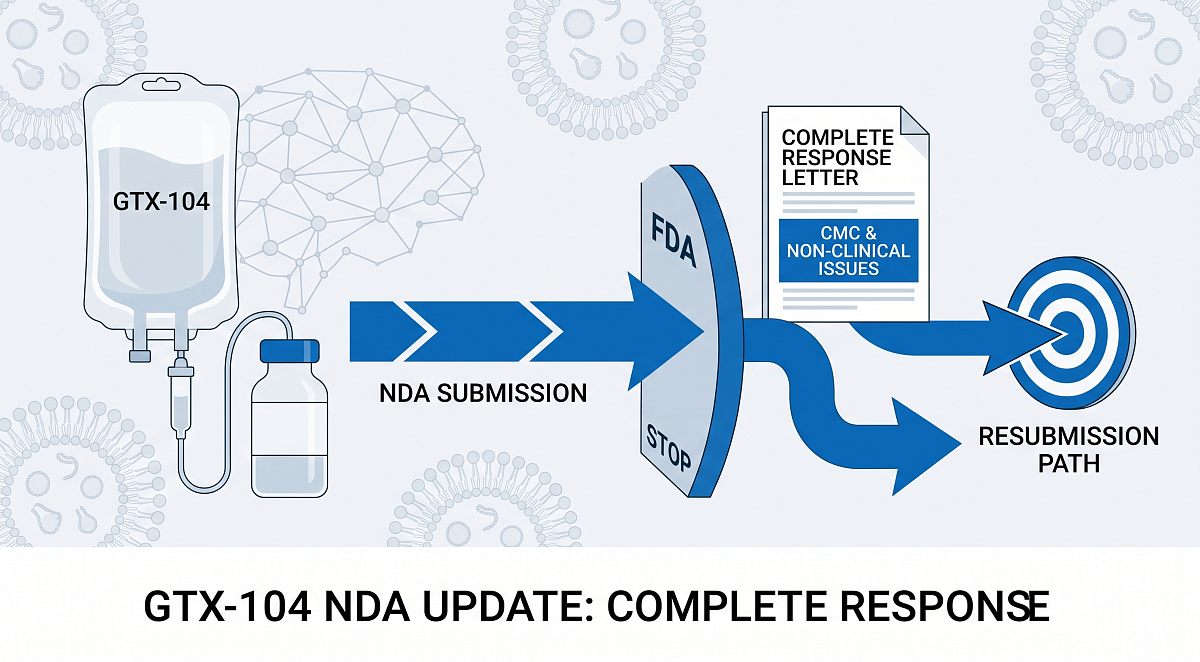
Nearly 10 months after the submission of a new drug application (NDA), the FDA has issued a complete response letter (CRL) for Grace Therapeutics’ GTx-104, an injectable formulation of Nimodipine, for the treatment of patients with aneurysmal subarachnoid hemorrhage (aSAH). The administration referenced items in the chemistry, manufacturing, and Controls (CMC) and non-clinical sections of the application.
The cited items are related to leachables data for product packaging, non-clinical product toxicology risk assessments, and product manufacturing deficiencies at Grace Therapeutics’ contract manufacturing organization. The company stated in a press release that it believes it can address the CRL in a resubmission of its NDA and intends to request a Type A meeting with the FDA to clarify the path forward.
“Potential FDA approval of our NDA for GTx-104 for the treatment of aSAH would represent the first meaningful innovation in the standard of care for these patients in more than 40 years,” stated Prashant Kohli, MD, chief executive officer of Grace Therapeutics. “We are confident in the robust data package supporting our NDA submission, and that the CMC issues identified by the FDA can be successfully addressed in our resubmission.”
About GTx-104 and Clinical Background
GTx-104 is administered intravenously and is designed to improve ease of use in the acute care setting, potentially eliminating the need for nasogastric tube administration in unconscious or dysphagic patients. The agent may reduce food effects, drug–drug interactions, and dosing variability. In addition, the company noted that GTx-104 may offer improved management of hypotension in patients with aSAH.
The NDA of GTx-104 was supported by findings from the phase 3 STRIVE-ON study (NCT05995405), a prospective, randomized, open-label study comparing GTx-104 to oral nimodipine in hospitalized patients. The trial met its primary end point, with a 19% reduction of at least 1 incidence of clinically significant hypotension observed in the GTx-104 group relative to oral nimodipine.