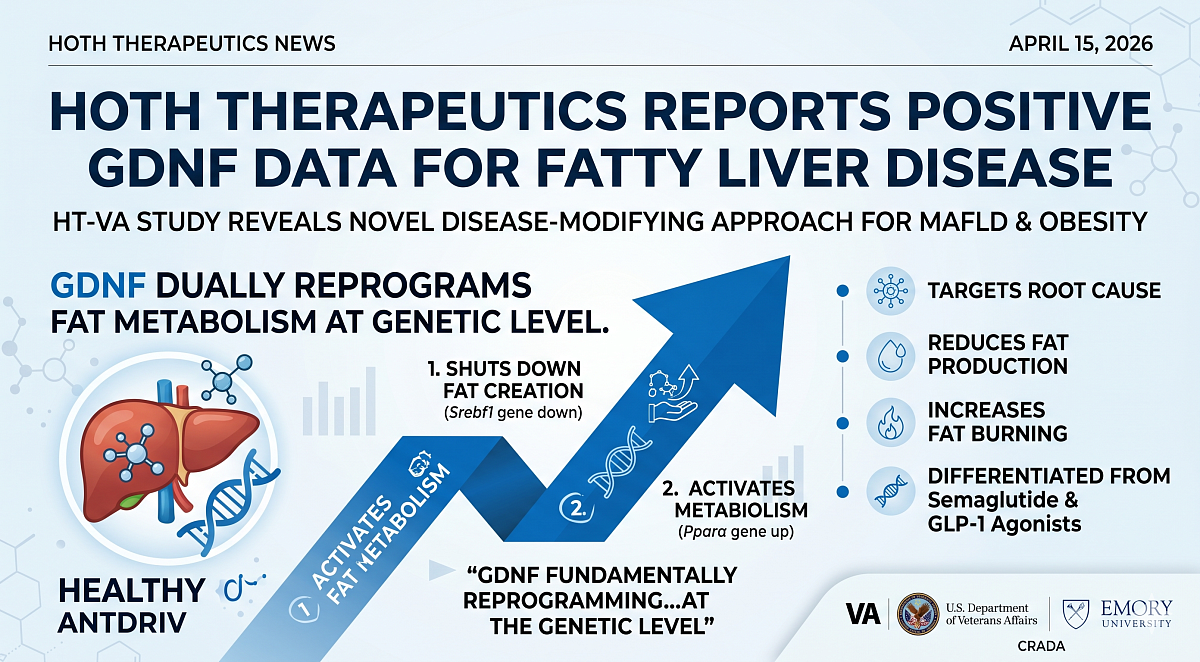
Hoth Therapeutics, a clinical-stage biopharmaceutical company, announced positive data from its HT-VA study, conducted under a CRADA with the U.S. Department of Veterans Affairs and Emory University. The data shows that parenteral GDNF (Glial Cell-Derived Neurotrophic Factor) directly reprograms liver fat metabolism at the genetic level, positioning GDNF as a potentially differentiated therapeutic approach targeting the root cause of fatty liver disease and metabolic dysfunction.
A Disease-Modifying Approach
Unlike existing therapies that primarily focus on weight loss, GDNF directly targets the biological mechanisms responsible for fat accumulation in the liver, demonstrating a dual mechanism of reducing fat production and increasing fat metabolism. This suggests GDNF may offer a disease-modifying approach for metabolic-associated fatty liver disease (MAFLD), obesity, and related metabolic disorders.
The HT-VA study evaluated the effects of parenteral GDNF in a diet-induced obesity and MAFLD model. Key observations include a statistically significant reduction in Srebf1, a key gene driving fat production in the liver, and increased expression of Pparα, a central regulator of fat metabolism and fat burning. GDNF also outperformed semaglutide in key gene expression markers tied to liver fat regulation.
“HT-VA represents a major milestone for Hoth as we expand into high-value metabolic indications. These results demonstrate that GDNF is not simply reducing fat, but fundamentally reprogramming how the body produces and metabolizes fat at the genetic level. The ability to shut down fat creation while activating fat metabolism differentiates GDNF from existing therapies, including GLP-1 agonists.”
— Robb Knie, Chief Executive Officer of Hoth Therapeutics
The Players
Hoth Therapeutics, Inc.
A clinical-stage biopharmaceutical company dedicated to developing innovative treatments to improve patient quality of life.
U.S. Department of Veterans Affairs
A government agency that collaborated with Hoth Therapeutics on the HT-VA study through a Cooperative Research and Development Agreement (CRADA).
Emory University
A research university that collaborated with Hoth Therapeutics on the HT-VA study through the CRADA.