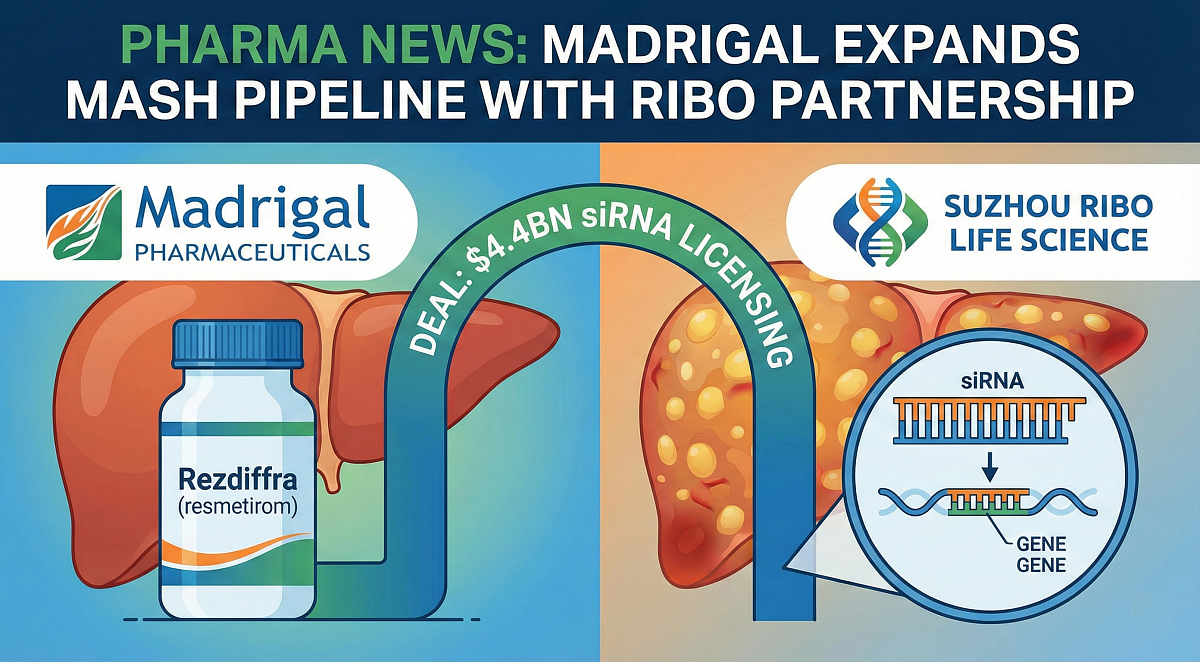
Madrigal Pharmaceuticals has inked a deal worth up to $4.4bn with Chinese biotech Suzhou Ribo Life Science and its subsidiary Ribocure Pharmaceuticals in a bid to expand offerings in the metabolic dysfunction-associated steatohepatitis (MASH) market.
The agreement will see Madrigal pay $60m upfront for the development and commercialisation rights to six of Ribo’s preclinical, silent interfering RNA (siRNA)-based MASH therapies. These therapies are designed to silence genes encoding proteins that drive the disease’s pathology.
Ribo will also be eligible to receive up to $4.4bn in development, regulatory, and commercial milestone payments, as well as potential royalties on sales involving these candidates. Through this deal, Madrigal can also choose to license some of Ribo’s other early-stage development programmes at a later date, including multiple bispecific siRNA candidates.
A key component of the deal with Ribo is the potential to combine the newly in-licensed candidates with Rezdiffra (resmetirom), which is Madrigal’s already approved therapy for MASH treatment.
At the core of Madrigal and Ribo’s agreement is the latter’s GalSTAR platform, which is designed to aid the development of RNA interference (RNAi) therapeutics targeting disease-causing genes involved in liver disease pathology.
Madrigal seeks to strengthen MASH presence
MASH, previously known as nonalcoholic steatohepatitis (NASH), is characterised by an accumulation of fat within the liver – triggering chronic inflammation within the organ. This can lead to scarring, liver cirrhosis, or hepatic cancer.
In 2024, Madrigal became the first company with a drug approved specifically for MASH in the form of thyroid hormone receptor beta (THR-β) agonist, Rezdiffra. Since then, the company has continued its MASH focus by acquiring Pfizer’s investigational candidate, ervogastat, for $50m, as well as two other preclinical assets.
Market Context: Rezdiffra brought in $287.3m in Q3 2025. GlobalData estimates that Rezdiffra’s sales will balloon to $6.9bn in 2032. However, the drug now faces competition from Novo Nordisk’s Wegovy (semaglutide), which gained MASH approval in August 2025.
GlobalData forecasts that the MASH market will reach a value of $20.3bn by 2032 – rising from 2022 at a compound annual growth rate (CAGR) of 38.2%.