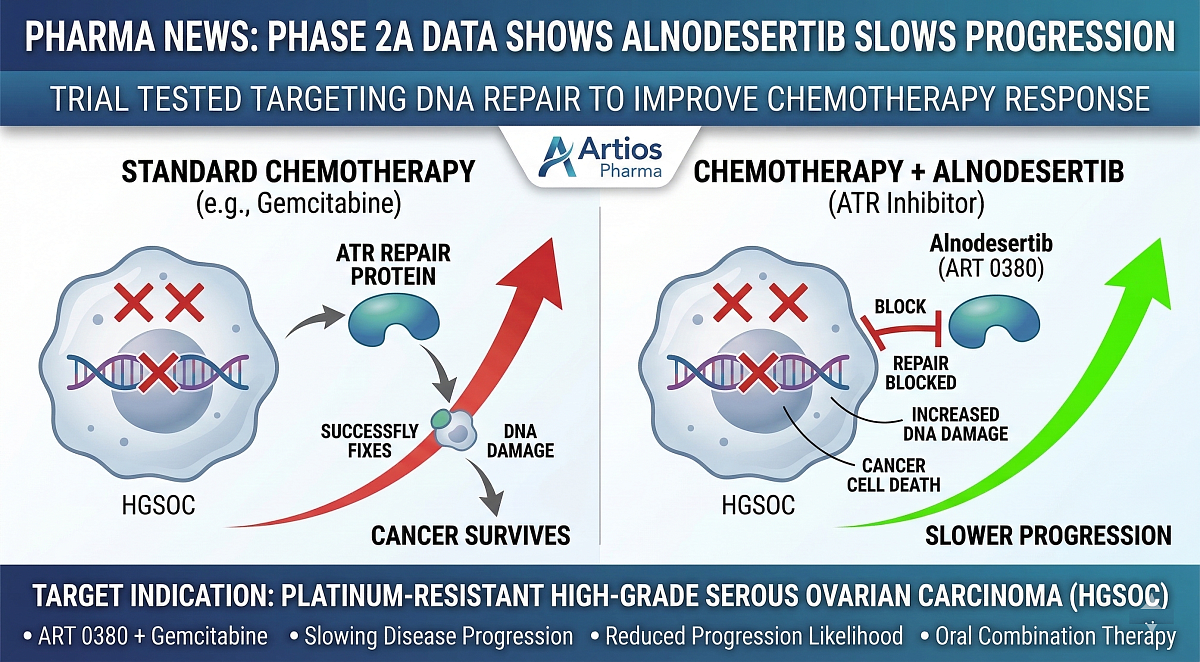
A low dose of alnodesertib (ART 0380) added to standard chemotherapy significantly reduced the likelihood of disease progression among people with platinum-resistant high-grade serous ovarian carcinoma (HGSOC) in an early clinical trial.
Findings from the Phase 2a study (NCT04657068) were announced by Artios Pharma, which is developing alnodesertib and sponsored the trial.
Why platinum-resistant ovarian cancer is difficult to treat
GSOC is a form of gynecological cancer marked by the uncontrolled growth of cells in or around the ovaries. First-line treatment usually involves platinum-based chemotherapies, but many patients eventually stop responding to these drugs.
Mechanism of Action: Targeting DNA Repair
For cells to divide, they must copy their DNA. When this process happens rapidly, as in fast-growing tumors like HGSOC, mistakes can occur, leading to DNA damage. This process, known as replication stress, can be deadly for cancer cells if they cannot repair the resulting DNA damage. Many cancer cells rely on a DNA repair protein called ATR (ataxia telangiectasia and Rad3-related) to help fix DNA damage caused by replication stress.
Alnodesertib, formerly known as ART 0380, is an oral therapy designed to block ATR activity, leaving cancer cells more vulnerable to DNA damage. The experimental medication is designed to work alongside existing gynecologic cancer treatments such as gemcitabine, a chemotherapy that damages cancer-cell DNA.