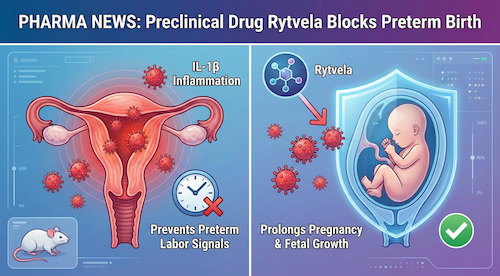
New research shows that Rytvela, an anti-inflammatory drug candidate, decreased premature birth and infant mortality when administered after the onset of preterm labor in a preclinical mouse model. The current standard treatment for preterm labor, Nifedipine, failed to confer similar benefits.
Preclinical Efficacy & Mechanism
The preclinical data, published in The Journal of Immunology, compared Rytvela, a new drug candidate, to Nifedipine, the most common drug for preterm labor in North America. Rytvela decreased rates of preterm birth by 40%, which prolonged gestation and fetal development.
Neonatal complications are highly associated with preterm birth, largely due to the underdevelopment of fetal organs. Rytvela prevented inflammation-induced neonatal tissue injury and promoted neonatal development, even when administered after exposure to inflammation.
Inflammation in the uterus and placenta is a major contributor to preterm labor, as it often sets off a series of inflammatory signals that induce labor. Rytvela targets the actions of a pro-inflammatory signal, called IL-1ß, to decrease inflammation. Unlike existing IL-1ß-targeting drugs, Rytvela avoids immune suppression, leaving the immune system intact to protect the mother and fetus.
Addressing an Unmet Need
Global Impact: Preterm birth affects 13.5 million births every year and is responsible for over 900,000 deaths annually.
Currently available treatments for women in preterm labor are drugs that try to stop or slow contractions. However, these drugs usually delay labor for less than 48 hours, which does not prevent preterm birth or allow the fetus more time to develop. Surviving infants may experience short- and long-term complications due to organs and systems not being fully developed at birth and exposure to dangerous inflammation.
Given the promising preclinical data, Rytvela has the potential to address many of these complications. The researchers are finalizing their preclinical research and preparing to proceed to clinical trials in people. The full research article is available in The Journal of Immunology.