Propanc Biopharma, Inc., a biopharmaceutical company focused on developing novel treatments for chronic diseases, including recurrent and metastatic cancer, today highlights the potential of its lead asset, PRP, as a novel therapeutic approach to the treatment and prevention of metastatic cancer from solid tumors, especially more aggressively spreading, less differentiated tumors, which offer a poor patient prognosis.
PRP May Outshine Traditional Therapies in a Booming $4.4 Billion Market
Pancreatic cancer is one of the deadliest cancers, with a five-year survival rate stuck at just 13% and no real progress has been made in recent years. To put that into perspective, overall cancer survival is 70%.
Standard treatments like chemotherapy (FOLFIRINOX or gemcitabine/nab-paclitaxel), targeted therapies (e.g., KRAS inhibitors), and emerging options (immunotherapies, tumor-treating fields like Optune Pax) extend life modestly but often bring harsh side effects, resistance, and limited success against this aggressive, metastasis-prone disease.
The Mechanism of PRP: Targeting Cancer Stem Cells
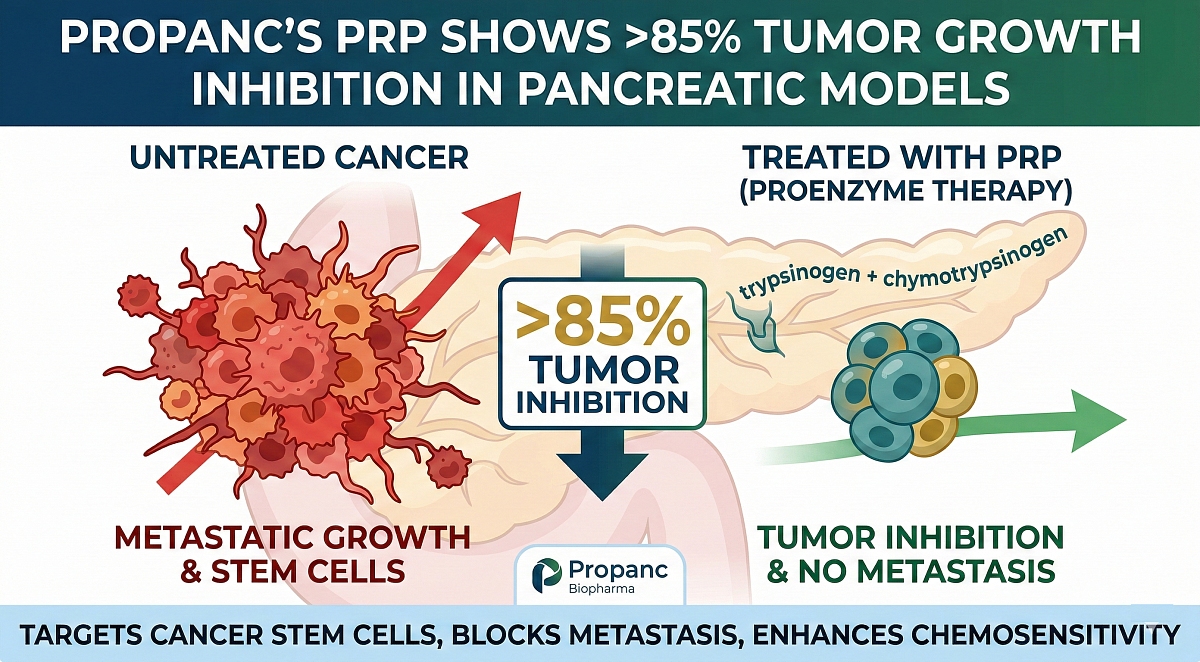
Enter Propanc's PRP — a promising investigational proenzyme therapy (trypsinogen + chymotrypsinogen in a 1:6 ratio) delivered intravenously. Unlike cytotoxic drugs that kill dividing cells broadly, PRP targets cancer stem cells, blocks metastasis by suppressing epithelial-mesenchymal transition (EMT), disrupts the tumor microenvironment, curbs angiogenesis, and boosts chemosensitivity—potentially making standard treatments more effective with far less toxicity.
Preclinical data shines: >85% tumor growth inhibition in pancreatic models, reduced fibrosis and resistance markers, and a gentler profile (no major side effects in limited prior human use). A small compassionate study (rectal version) extended survival from ~5.6 to 9 months in advanced cases.
PRP vs. Current Treatment Options
How PRP Compares:
- Chemo: PRP could sensitize resistant tumors and cut doses/side effects.
- Targeted drugs: Broader attack on stem cells and spread, not just single mutations.
- Immunotherapy: May warm up “cold” pancreatic tumors by remodeling the microenvironment.
Market Outlook and Future Trials
According to industry sources, the global pancreatic cancer treatment market is valued at ~$4.42 billion in 2026 and projected to explode to $14.43 billion by 2034 (CAGR ~16%), fueled by rising cases and demand for better options.
Propanc is gearing up for a Phase 1b First-In-Human trial in 2026 (30–40 advanced solid tumor patients), backed by fresh funding ($100M facility), new patents, and FDA Orphan Drug status for pancreatic cancer.