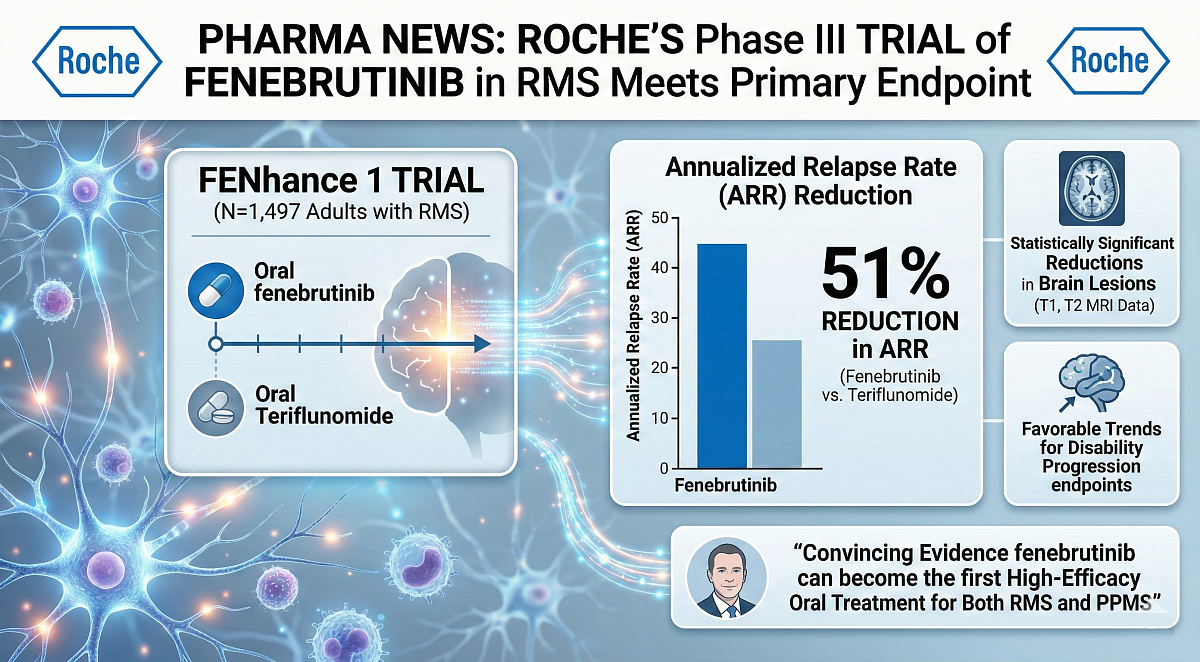
Roche has reported that the pivotal Phase III FENhance 1 trial of its investigational Bruton’s tyrosine kinase (BTK) inhibitor, fenebrutinib, in relapsing multiple sclerosis (RMS), achieved the primary endpoint.
The trial results showed a reduction of 51% in annualised relapse rate (ARR) when compared to teriflunomide after a minimum of 96 weeks of treatment. This finding aligns with results from FENhance 2, which reported a 59% reduction. Statistically significant reductions in brain lesions were observed across both studies, with all progression endpoints showing favourable trends for fenebrutinib.
Trial Design and Endpoints
The randomised, multi-centre, double-dummy, parallel-group, double-blind FENhance 1 and 2 trials involved 1,497 adults with RMS. Participants received either oral fenebrutinib twice daily or oral teriflunomide once daily for at least 96 weeks. After the double-blind period, patients were offered an open-label extension phase with fenebrutinib.
Study Goals and Metrics:
- Primary endpoint: The annualised relapse rate (ARR).
- Secondary goals encompass:
- The overall count of T1-gadolinium-enhancing magnetic resonance imaging (MRI) lesions.
- The sum of new or enlarged T2-weighted MRI lesions.
- The duration until the start of a 12-week composite confirmed disability progression (cCDP12).
- The time to a 24-week cCDP (cCDP24).
FENhance 1 represents the conclusive study readout of the fenebrutinib pivotal clinical development programme for MS. This follows the favourable outcomes previously observed in FENhance 2 for RMS and in FENtrepid for primary progressive multiple sclerosis (PPMS).
Recent Developments in Other Pipelines
In related clinical news beyond neurology, in January 2026, Roche reported positive top line results from its Phase II trial in obesity, where a once-weekly subcutaneous injection of CT-388 resulted in significant weight loss.