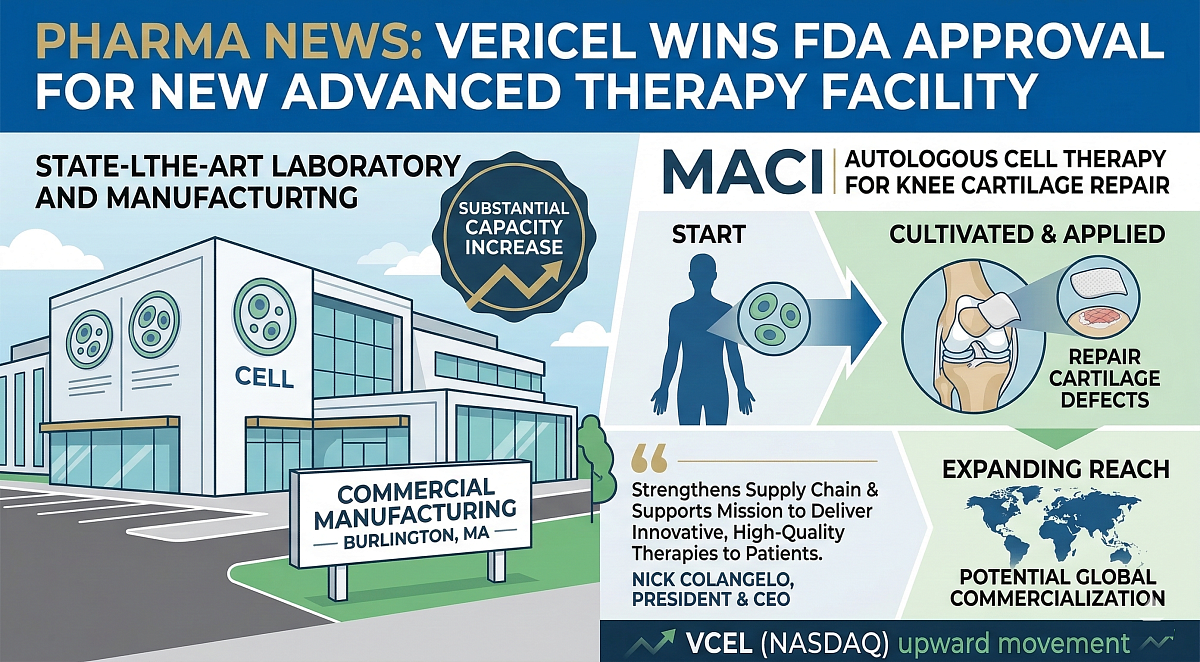
(RTTNews) — Vericel Corporation (VCEL) announced that the U.S. Food and Drug Administration (FDA) has approved commercial manufacturing of MACI (autologous cultured chondrocytes on porcine collagen membrane) at its new state-of-the-art cell therapy facility in Burlington, Massachusetts.
The approval allows Vericel to begin production in the second quarter of 2026, substantially increasing capacity to meet the growing clinical demand for MACI, its flagship autologous cell therapy for cartilage repair in the knee. The company also noted that the expanded facility positions it to potentially commercialize MACI outside of the United States.
What is MACI?
Autologous cultured chondrocytes on porcine collagen membrane (MACI) is a specialized treatment designed to correct cartilage defects in the knee. By utilizing the patient's own cartilage cells, the therapy aims to restore the knee's structural integrity and function.
Expanding Portfolio and Market Reach
Vericel currently markets three advanced therapies in the U.S. market: MACI for targeted cartilage repair, Epicel for severe burn treatment, and NexoBrid for eschar removal in burn patients. The new facility approval marks a significant regulatory and operational milestone as Vericel continues to scale its overall business and production capabilities.