Factor XIa Compounds Library
Description
ChemDiv’s library of small molecule compounds targeting factor XIa comprises 6,500 entries.
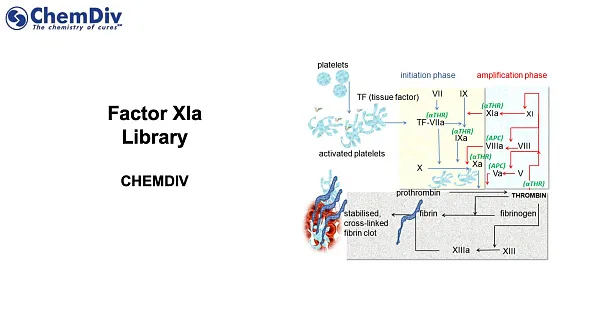
Currently, all FDA-approved anticoagulants come with an extensive list of side effects, most of which are related to bleeding. The primary target for these drugs is a coagulation cascade, which consists of two major pathways:
● Extrinsic pathway, initiated by endothelial damage or hypoxia.
● Intrinsic pathway, triggered by damage to blood vessels.
There is clinical evidence suggesting that selective inhibition of the intrinsic pathway could reduce the risk of bleeding. Factor XIa (FXIa, also known as plasma thromboplastin antecedent) is a serine protease in the intrinsic pathway that plays a crucial role in the amplification of the procoagulant signal. Consequently, it emerges as an attractive target for treating thrombotic disorders [1].
Our library, comprising carefully selected virtual hits with marked affinity to Factor XIa, offers a solid foundation for developing novel anticoagulants. Their mechanism of action is specifically focused on inhibiting Factor XIa, potentially offering a safer alternative to current treatments.
By focusing on compounds that selectively inhibit Factor XIa, our library addresses the major challenge of reducing bleeding risks associated with current anticoagulants. Researchers can utilize this collection to rapidly screen and identify promising candidates that exhibit specific inhibitory effects on Factor XIa, accelerating the early stages of drug development. The high degree of specificity in these virtual hits enhances the likelihood of discovering effective anticoagulant agents with fewer side effects, paving the way for the creation of next-generation anticoagulant therapies that offer improved safety profiles for patients.
The library was created through a meticulous development process that began with the removal of compounds possessing undesirable properties, ensuring a high-quality starting point [2,3]. Advanced machine learning (ML) models, complemented by ICM docking techniques, guided the virtual screening process. This approach allowed for the efficient identification of compounds with potential efficacy based on their interaction with specific biological targets. The ML models used in the screening process demonstrated an accuracy of 0.86, indicating a high level of reliability in predicting compounds' biological activities and interactions. Initially, the ML-based selection process yielded approximately 200,000 compounds. This number was further refined to around 25,000 compounds, specifically those with ICM docking scores greater than 23, ensuring that only the most promising candidates were included. The library emphasizes structural diversity, achieved through hierarchical clustering and min-max algorithms, along with dice similarity metrics. This diversity is crucial for exploring a wide range of potential biological activities and mechanisms of action.
Product Details
Set of small-molecule compounds that can target Factor XIaLibrary Composition
Set of small-molecule compounds that can target Factor XIaPublications
[1] Corte JR, Fang T, Osuna H, Pinto DJ, Rossi KA, Myers JE Jr, Sheriff S, Lou Z, Zheng JJ, Harper TW, Bozarth JM, Wu Y, Luettgen JM, Seiffert DA, Decicco CP, Wexler RR, Quan ML. Structure-Based Design of Macrocyclic Factor XIa Inhibitors: Discovery of the Macrocyclic Amide Linker. J Med Chem. 2017 Feb 9;60(3):1060-1075. doi: 10.1021/acs.jmedchem.6b01460.[2] Baell JB, Holloway GA. New substructure filters for removal of pan assay interference compounds (PAINS) from screening libraries and for their exclusion in bioassays. J Med Chem. 2010; 53(7):2719-40. doi: 10.1021/jm901137j.
[3] Sushko I, Salmina E, Potemkin VA, Poda G, Tetko IV. ToxAlerts: a Web server of structural alerts for toxic chemicals and compounds with potential adverse reactions. J Chem Inf Model. 2012; 52(8):2310-6. doi: 10.1021/ci300245q.