C-Met Library
C-Met Inhibitors Library
The ChemDiv’s library of c-Met inhibitors contains 16,000 small molecule compounds that inhibit the enzymatic activity of the c-Met tyrosine kinase, the receptor of hepatocyte growth factor/scatter factor (HGF/SF).
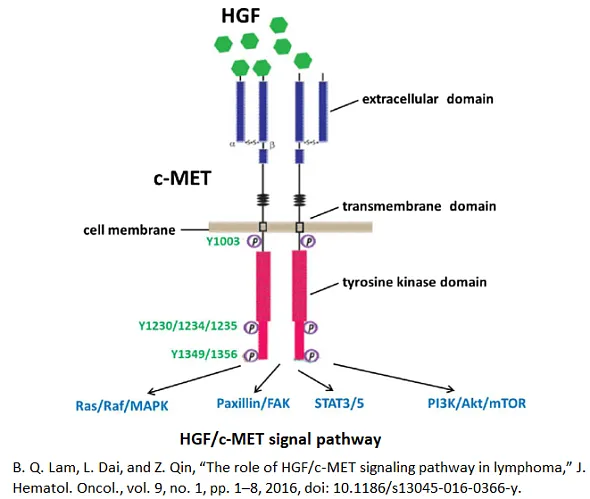
Receptor tyrosine kinases (RTKs) are involved in regulating numerous critical cellular processes in mammalian development mechanisms, cellular functionality, and tissue homeostasis. The c-MET proto-oncogene encodes the c-MET tyrosine kinase protein, a cell surface receptor prominently expressed in the epithelial cells of various organs, in particular, liver, pancreas, prostate, kidney, muscle, and bone marrow, which can be noted during both embryogenesis and adulthood. The ligand for c-MET is hepatocyte growth factor (HGF), which functions as a pleiotropic factor and cytokine, stimulating cell proliferation, survival, motility, scattering, differentiation, and morphogenesis. The interaction between c-MET and HGF can initiate the activation of several signaling pathways, such as PI3K and mTOR. Given the integral role of RTKs in normal physiological processes, the dysregulation of specific RTKs, including c-MET, has been implicated in the onset and progression of various cancers. Enhanced expression of c-MET and HGF has been found in tumor biopsies from a majority of solid tumors, and c-MET signaling is prevalent across a wide spectrum of human malignancies [1].
c-MET inhibitors have gained significant attention in drug discovery, particularly in the field of oncology, due to their role in targeting the c-MET/HGF signaling pathway. This pathway is critical in cellular processes such as proliferation, survival, and migration, and its dysregulation is implicated in the development and progression of various cancers. By inhibiting c-MET, these drugs aim to block the aberrant signaling responsible for tumor growth and metastasis. The development of small molecule c-MET inhibitors offers a targeted therapeutic approach, especially for cancers that exhibit overexpression or mutation of the c-MET gene. Additionally, c-MET inhibitors are being explored in combination with other cancer therapies to overcome resistance mechanisms and enhance treatment efficacy, showcasing their potential in advancing personalized medicine for cancer patients.
The major portion of drug compounds being developed todat target the receptor directly. Small molecule inhibitors can function as either ATP-competitive or non-competitive agents. ATP-competitive inhibitors, while potent, tend to have lower specificity due to their potential to inhibit other kinases, given their interaction with the ATP-binding pocket common to many kinases. In contrast, non-competitive inhibitors are often more specific, as they typically interact with allosteric sites on the receptor rather than the ATP pocket. These sites are unique to each kinase, thus, reducing the risk of off-target effects. Checmila interaction with those allosteric sites induces conformational changes in the active site, thereby modulating the binding of the ligand HGF. An example of an ATP-competitive drug is cabozantinib, which effectively inhibits the c-MET receptor and demonstrates significant antitumor activity [2].
References
[1] S. L. Organ and M. S. Tsao, “An overview of the c-MET signaling pathway,” Therapeutic Advances in Medical Oncology, vol. 3, no. 1. SAGE Publications, pp. S7–S19, 2011, doi: 10.1177/1758834011422556.
[2] A. Fasolo, C. Sessa, L. Gianni, and M. Broggini, “Seminars in clinical pharmacology: An introduction to met inhibitors for the medical oncologist,” Ann. Oncol., vol. 24, no. 1, pp. 14–20, 2013, doi: 10.1093/annonc/mds520.