Gilead Announces U.S. FDA Priority Review Designation for Sofosbuvir/Velpatasvir for Treatment of All Genotypes of Chronic Hepatitis C Infection
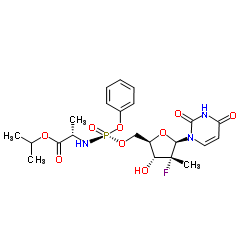
Sofosbuvir
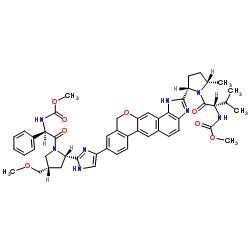
Velpatasvir
FOSTER CITY, Calif.--(BUSINESS WIRE)--Jan. 4, 2016-- Gilead Sciences, Inc. (Nasdaq: GILD) today announced that the U.S. Food and Drug Administration (FDA) has granted priority review to the company’s New Drug Application (NDA) for an investigational, once-daily fixed-dose combination of the nucleotide analog polymerase inhibitor sofosbuvir (SOF), approved as Sovaldi® in December 2013, and velpatasvir (VEL), an investigational pan-genotypic NS5A inhibitor, for the treatment of chronic genotype 1-6 hepatitis C virus (HCV) infection. Gilead filed the NDA for SOF/VEL on October 28, 2015, and FDA has set a target action date under the Prescription Drug User Fee Act (PDUFA) of June 28, 2016.
The FDA has assigned SOF/VEL a Breakthrough Therapy designation, which is granted to investigational medicines that may offer major advances in treatment over existing options. The NDA for SOF/VEL is supported by data from four Phase 3 ASTRAL trials, which evaluated the fixed-dose combination in hepatitis C genotypes 1-6. A marketing application for SOF/VEL is also under review in the European Union, and was validated by the European Medicines Agency (EMA) in December. The SOF/VEL fixed-dose combination is an investigational product and its safety and efficacy have not been established.
About Gilead Sciences
Gilead Sciences is a biopharmaceutical company that discovers, develops and commercializes innovative therapeutics in areas of unmet medical need. The company’s mission is to advance the care of patients suffering from life-threatening diseases worldwide. Gilead has operations in more than 30 countries worldwide, with headquarters in Foster City, California.
Source: http://www.gilead.com