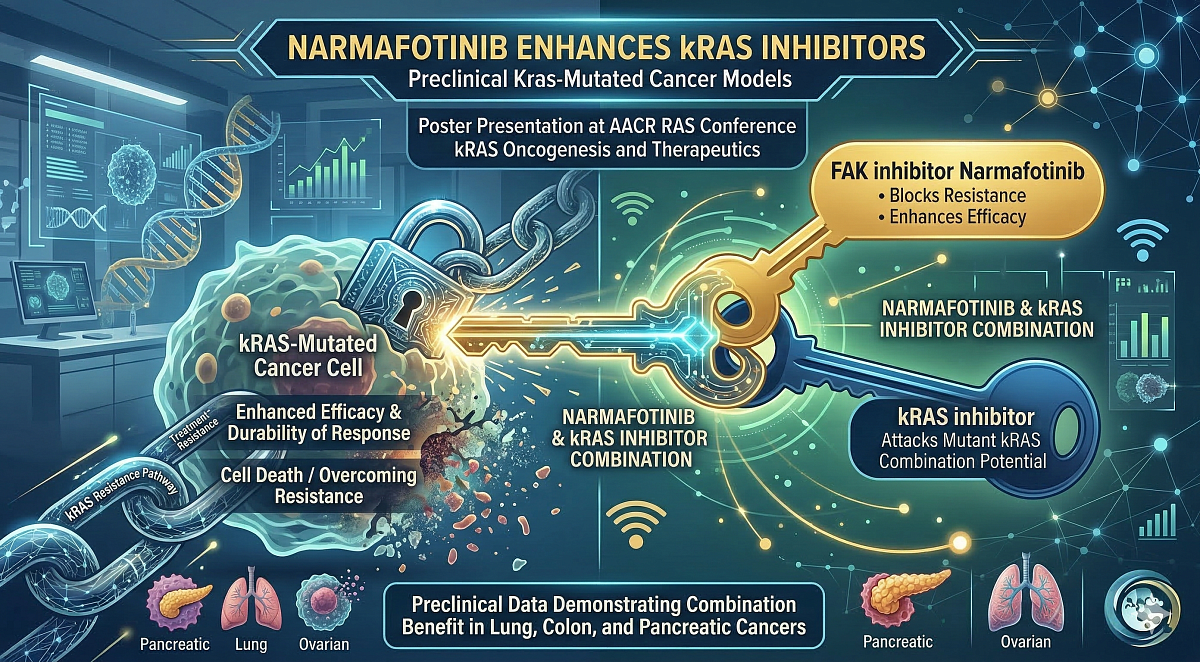
Poster Presentation Demonstrating Combination Benefit of Narmafotinib in Preclinical Kras-Mutated Cancer Models
Amplia Therapeutics Limited announces that compelling data describing new clinical opportunities for its lead drug narmafotinib was presented at the AACR Special Conference in Cancer Research: RAS Oncogenesis and Therapeutics in Los Angeles, California on Friday March 6.
Amplia’s lead drug narmafotinib enhances the activity of kRAS inhibitors in multiple preclinical cancer models presented at US conference. Data showing narmafotinib activity in preclinical models of pancreatic cancer, lung cancer and ovarian cancer are presented. The development of kRAS inhibitors for different types of solid tumors is currently an area of intense global activity.
The poster presentation discloses preclinical data demonstrating that the Company’s best-in-class FAK inhibitor narmafotinib enhances the activity of a new class of drugs called kRAS inhibitors in various models of cancer. In particular, the data indicates that narmafotinib blocks resistance pathways that can emerge with kRAS inhibitor treatment, thereby enhancing efficacy and durability of response.
Inhibitors of mutant kRAS proteins are an exciting new class of drug in development for the treatment of lung, colon and pancreatic cancer, amongst others. There are currently over 50 different kRAS inhibitors undergoing clinical studies across the globe. Despite promising mid-stage clinical data, however, side-effects of these drugs can be significant and treatment-emergent resistance is commonplace.
"We are excited to present our research findings at this specialist conference focused on RAS inhibition in cancer. We believe there is significant clinical potential in combining narmafotinib with kRAS inhibitors and will be discussing our findings with pharma and biotech companies actively working in this space."