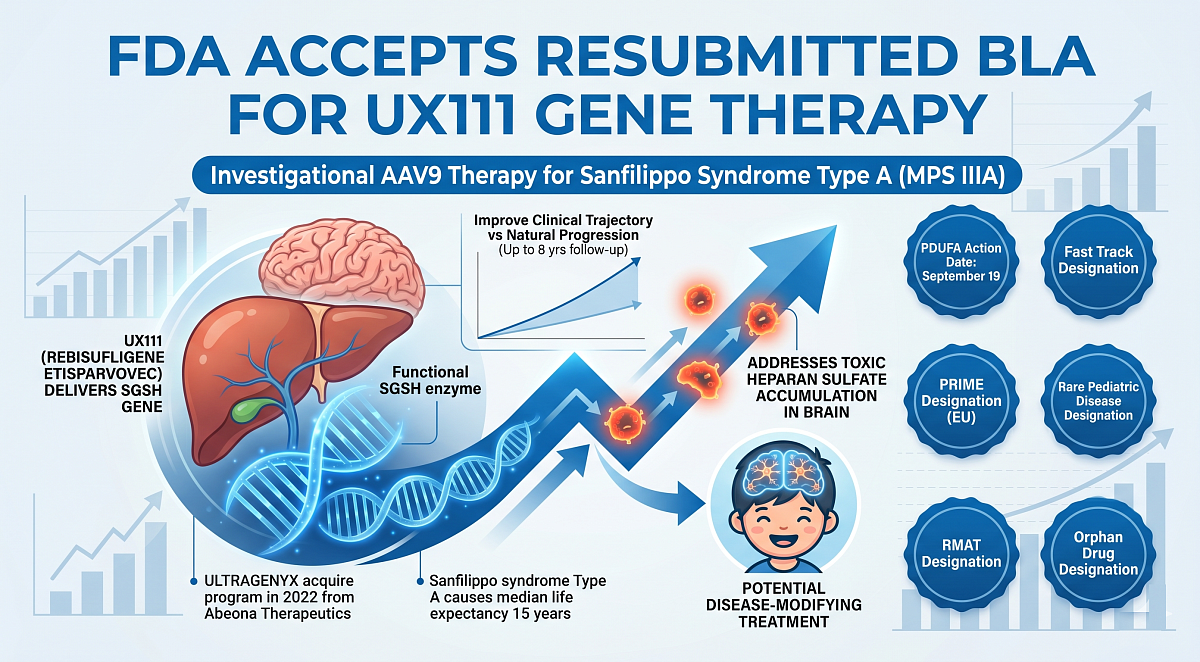
On April 2, Ultragenyx announced that the US FDA accepted the resubmitted Biologics License Application (BLA) for UX111, an investigational AAV9 gene therapy for Sanfilippo syndrome Type A (MPS IIIA). The FDA set a PDUFA action date of September 19 for its decision. If approved, UX111 would become the first available treatment for this rare, fatal genetic disorder, which causes progressive and irreversible neurodegeneration in young children.
How UX111 (Rebisufligene Etisparvovec) Works
The therapy, also known as rebisufligene etisparvovec, is a one-time intravenous infusion designed to deliver a functional copy of the SGSH gene. This addresses the underlying enzyme deficiency responsible for the toxic accumulation of heparan sulfate in the brain. Clinical data included in the BLA, spanning up to 8 years of follow-up, demonstrate a durable treatment effect and clinical improvement compared to the natural progression of the disease. The FDA previously acknowledged the robustness of the neurodevelopmental and biomarker data during the prior review cycle.
Addressing Sanfilippo Syndrome Type A
Sanfilippo syndrome Type A is caused by a lack of the sulfamidase enzyme, leading to a median life expectancy of only 15 years. Ultragenyx’s UX111 received multiple high-level regulatory designations, including Fast Track and Rare Pediatric Disease status. If granted accelerated approval, the therapy will be manufactured at specialized facilities in Ohio and Massachusetts, providing a critical new option for a community currently without any approved disease-modifying treatments.
The UX111 program, which Ultragenyx acquired from Abeona Therapeutics in 2022, has received Regenerative Medicine Advanced Therapy, Fast Track, Rare Pediatric Disease, and Orphan Drug designations in the United States and PRIME and Orphan medicinal product designations in the EU.
While Ultragenyx has its work cut out, it could be another win for the gene therapy community.
About Ultragenyx
Ultragenyx is a biotech company that is focused on bringing novel products to patients for the treatment of serious rare and ultra-rare genetic diseases.