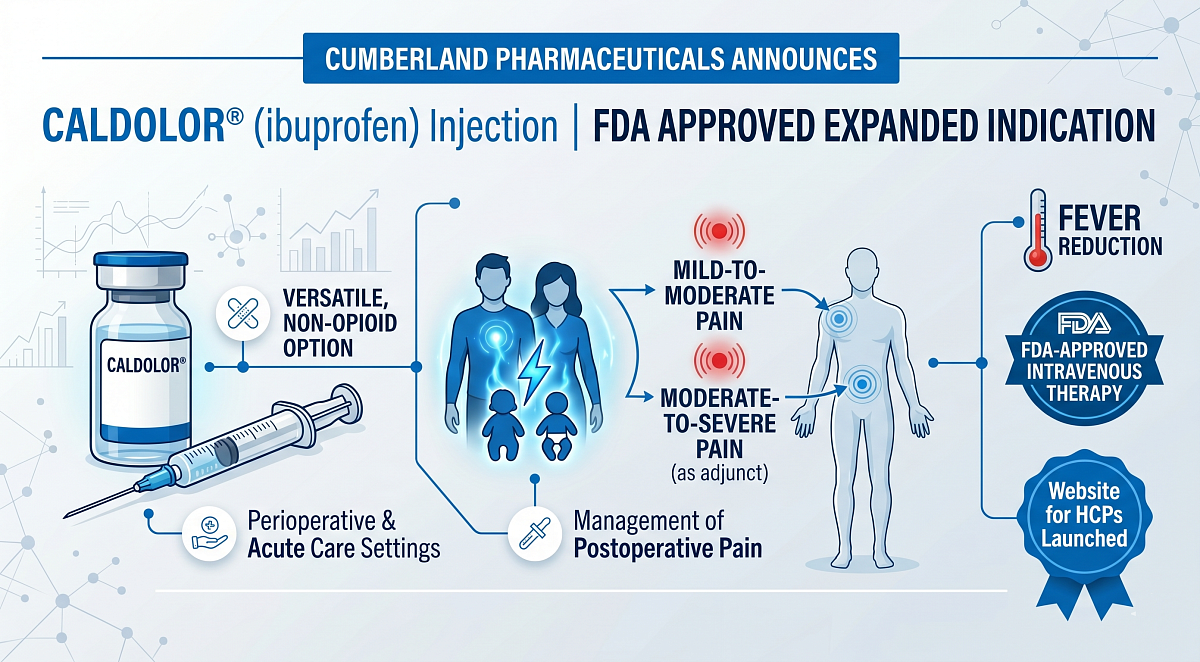
Cumberland Pharmaceuticals Inc., a specialty pharmaceutical company, announced it has received approval from the US Food and Drug Administration (FDA) for an expanded indication for its Caldolor (ibuprofen) injection product. The indication now includes the management of postoperative pain, and the approval coincides with the launch of a newly designed website for healthcare professionals highlighting this advancement and the evolving role of non-opioid pain management.
Expanded Indication Details
With this update, Caldolor is indicated for use in adult and paediatric patients ages 3 months and older for:
- Management of mild to moderate pain, including postoperative pain
- Management of moderate to severe pain, including postoperative pain, as an adjunct to opioid analgesics
- Reduction of fever
This expanded labelling reinforces Caldolor's role as a versatile, non-opioid intravenous analgesic option for use in perioperative and acute care settings.
New Resources for Healthcare Professionals
The newly launched Caldolor website aligns with this expanded indication, featuring dedicated content on postoperative pain management and the importance of multimodal, opioid-sparing approaches.
Postoperative pain remains a significant challenge across surgical setting, with a growing emphasis on reducing opioid exposure while maintaining effective pain control. The updated website provides healthcare professionals with resources that focus on:
- The role of IV non-opioid options like Caldolor in perioperative care
- Strategies to support opioid stewardship amid the ongoing opioid crisis
- Clinical data supporting safe and effective pain and fever management with Caldolor
In addition to featuring the new postoperative pain indication, the website offers comprehensive information on safety, dosing, efficacy, and access with J-Code reimbursement support.
The site reinforces Caldolor as "A Safe and Effective Non-Opioid Pain Management Solution."
Important Safety Information & Indications
Caldolor is indicated in adults and paediatric patients for the management of mild to moderate pain and management of moderate to severe pain, including postoperative pain management, as an adjunct to opioid analgesics, as well as the reduction of fever. It was the first FDA-approved intravenous therapy for fever. Caldolor is contraindicated in patients with known hypersensitivity to ibuprofen or other non-steroidal anti-inflammatory drugs (NSAIDs) and patients with a history of asthma or other allergic type reactions after taking aspirin or other NSAIDs. Caldolor is contraindicated for use during the peri-operative period in the setting of coronary artery bypass graft (CABG) surgery.
About Cumberland Pharmaceuticals Inc.
Cumberland Pharmaceuticals Inc. is the largest biopharmaceutical company founded and headquartered in Tennessee and is focused on providing unique products that improve the quality of patient care. The company develops, acquires, and commercializes products for the hospital acute care, gastroenterology and oncology market segments.