FDA Grants Fast Track to BBO-11818 for Advanced KRAS-Mutant Pancreatic
BBOT Granted U.S. FDA Fast Track Designation for BBO-11818
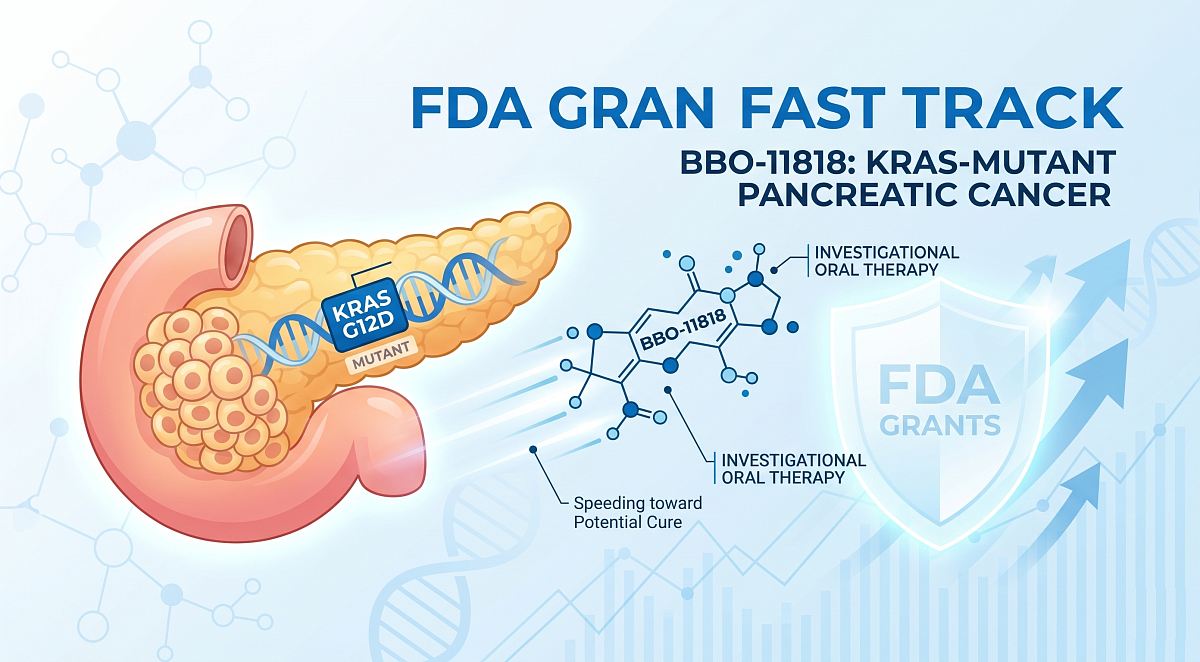
BridgeBio Oncology Therapeutics, Inc. (BBOT), a pioneering clinical-stage biopharmaceutical company focused extensively on RAS-pathway malignancies, today proudly announced that the U.S. Food and Drug Administration (FDA) has officially granted Fast Track designation to BBO-11818. This designation specifically targets the critical treatment of adult patients diagnosed with advanced KRAS-mutant pancreatic ductal adenocarcinoma.
Addressing a Critical Unmet Need
"Receiving the highly coveted Fast Track designation from the FDA for BBO-11818 in KRAS-mutant pancreatic ductal adenocarcinoma directly reflects the profound importance and pressing urgency of successfully accelerating the clinical development of our innovative pan-KRAS inhibitor in this extraordinarily serious disease," stated Yong Ben, MD, Chief Medical and Development Officer of BBOT.
"Pancreatic cancer tragically remains one of the absolute most difficult-to-treat malignancies globally. KRAS mutations are actively present in the vast majority of diagnosed cases, yet patients historically have had remarkably few targeted therapeutic options available to them. This crucial FDA designation will significantly help us collaborate closely and rapidly with the agency to strategically advance BBO-11818 as safely and efficiently as entirely possible for vulnerable patients who urgently need new, effective options."
Clinical Progress and Drug Profile
This major designation closely follows highly promising preliminary data, first publicly released in January 2026, which clearly demonstrated that BBO-11818 monotherapy achieved a confirmed, meaningful partial response in pancreatic cancer patients. In addition to this milestone, broad anti-tumor activity was consistently observed across various dose levels and diverse tumor types, featuring marked tumor reductions at higher dose levels alongside a generally favorable and highly differentiated safety profile noted during strict dose escalation phases.
While existing KRASG12C inhibitors have certainly demonstrated promising clinical efficacy recently, there undeniably remains a clear, glaring unmet medical need for advanced therapies targeting aggressive cancers that persistently carry other complex KRAS mutations, such as KRASG12D and KRASG12V. BBO-11818 was meticulously developed specifically to address this exact gap in care. It is ingeniously designed as a highly potent pan-KRAS inhibitor possessing strong, specific binding affinity for KRAS, exceptionally high selectivity over HRAS and NRAS, and the unique ability to achieve high, sustained levels of KRAS inhibition in both the active ON and inactive OFF states.
Future Trials and Company Mission
BBO-11818 is currently being thoroughly evaluated as a powerful monotherapy, in strategic combination with established standard-of-care therapies, or acting alongside BBOT’s proprietary RAS:PI3Kα breaker, BBO-10203. It is actively advancing in the Phase 1 KONQUER-101 trial focusing on subjects battling locally advanced unresectable or fully metastatic KRAS-mutant solid tumors. The FDA's Fast Track designation is fundamentally intended to help rapidly advance the vital development and comprehensive review process for promising new therapeutic candidates targeting serious conditions that clearly fill a massive unmet medical need.
About BBOT: BBOT is an advanced clinical-stage biopharmaceutical company relentlessly advancing a next-generation pipeline of entirely novel small molecule therapeutics specifically targeting deadly RAS and PI3Kα malignancies. The company’s ultimate goal is decisively improving long-term outcomes for patients afflicted with cancers directly driven by the two absolute most prevalent oncogenes identified in human tumors.