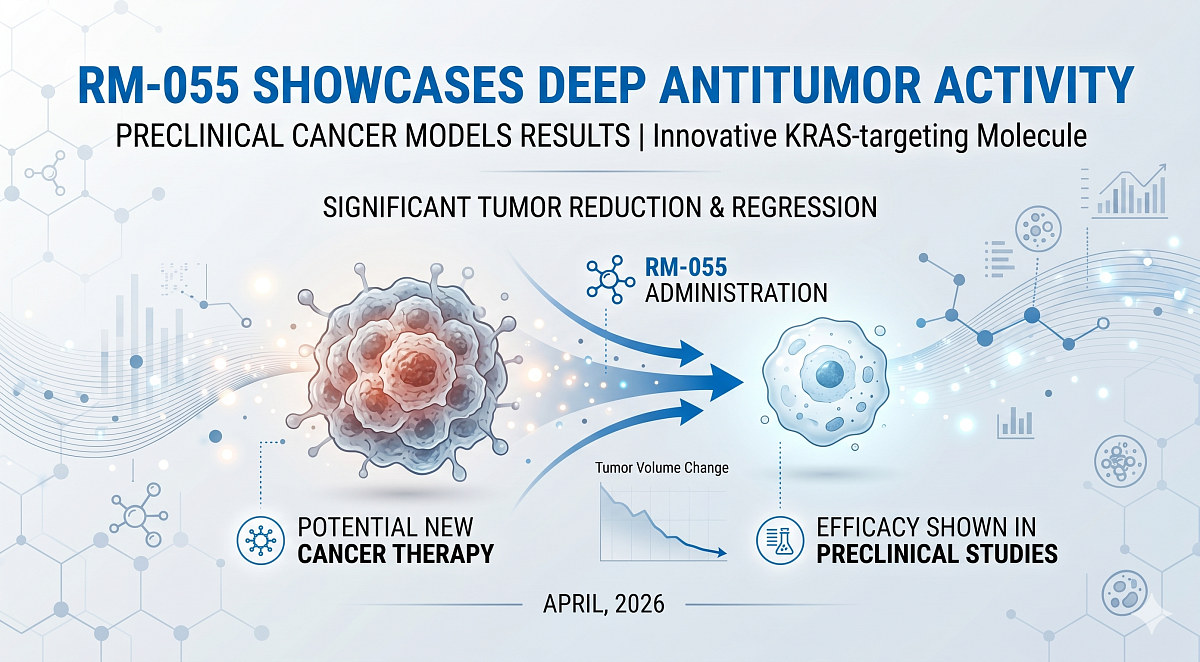
RM-055 Highlights Deep Antitumor Activity in Preclinical Cancer Models
Data Highlight RM-055's Ability to Overcome Resistance Across Preclinical Models
Revolution Medicines, Inc., a leading late-stage clinical oncology company focused on developing targeted therapies for patients heavily afflicted with RAS-addicted cancers, today announced compelling preclinical data. The findings highlight an innovative new class of mutant-targeted catalytic RAS(ON) inhibitors.
Innovative Mechanism of Action
These highly specialized catalytic inhibitors are explicitly designed to promote the direct conversion of mutant RAS from its active RAS(ON) state back to the inactive RAS(OFF) state, thereby closely mimicking the physiological regulation typically seen in wild type RAS. This unique mechanism represents a highly differentiated approach to the comprehensive inhibition of oncogenic RAS signaling, fundamentally aiming to address emerging tumor resistance mechanisms and significantly extend the durability of current RAS-targeted therapeutic options.
Comprehensive results of this study will be officially presented during a highly anticipated mini-symposium at the American Association for Cancer Research (AACR) Annual Meeting on April 21, 2026 (Abstract #6782).
Insights from Leadership
“Additional, robust strategies are urgently needed to effectively counter emergent resistance to RAS inhibitors and further extend tangible clinical benefit for patients currently battling RAS mutant cancers,” stated Jan Smith, Ph.D., chief scientific officer at Revolution Medicines. “Using our advanced cyclophilin A tri-complex platform, we have successfully discovered a completely new class of mutant-targeted RAS(ON) catalytic inhibitors designed specifically to stimulate the GTPase activity of mutant RAS variants—a long-sought, critical goal of the global RAS research community. In rigorous preclinical models, RM-055, an oral compound utilizing this exact mechanism as the primary driver of RAS pathway inhibition, consistently drove deep and durable tumor regressions across multiple tumor types and successfully overcame entrenched resistance to prior RAS inhibition.”
Preclinical Results and RM-055 Efficacy
At demonstrably well-tolerated doses, RM-055 demonstrated robust and highly durable antitumor activity across multiple KRAS G12 mutant xenograft models, specifically targeting pancreatic ductal adenocarcinoma, non-small cell lung cancer, and colorectal cancer. Notably, aggressive tumors that had previously escaped conventional RAS inhibitor treatment remained highly sensitive to RM-055. The compound drove deep and durable regressions, strongly warranting further clinical investigation of its vast potential to counter emergent drug resistance and extend patient clinical benefit.
Mutant RAS variants causing cancer are notoriously relatively insensitive to the natural GTPase-activating proteins (GAPs) that normally induce physiological inactivation of wild type RAS by stimulating the essential hydrolysis of RAS-GTP to RAS-GDP. Similar to the vital natural GAPs, RM-055 directly accelerates the critical hydrolysis of mutant RAS-GTP to RAS-GDP, effectively converting oncogenic RAS from its active RAS(ON) state to a safe, inactive RAS(OFF) state. Remarkably, a single cyclophilin A:RM-055 binary complex is fully capable of inactivating multiple mutant RAS proteins sequentially.
Future Potential and Company Overview
In extensive preclinical studies, RM-055 significantly reduced dangerous RAS-GTP levels in cells, directly leading to powerful inhibition of downstream RAS signaling and halting tumor cell proliferation. In vivo, RM-055 preferentially and selectively suppressed RAS pathway activation within KRAS G12 mutant tumors over healthy normal tissues. This highly mutant-targeted activity, combined with a dramatically reduced impact on wild-type RAS in normal tissues, strongly suggests the distinct potential for an optimally enhanced therapeutic window and significantly increased flexibility for future combination treatment approaches. Moreover, this approach may consistently enable durable pathway suppression even in tumors demonstrating increased RAS signaling, which is a very common mechanism of clinical acquired resistance to standard RAS inhibition.
Revolution Medicines continues to advance its R&D pipeline comprising RAS(ON) inhibitors like daraxonrasib (RMC-6236), elironrasib (RMC-6291), zoldonrasib (RMC-9805), and RMC-5127, all currently in clinical development to suppress diverse oncogenic variants of RAS proteins.