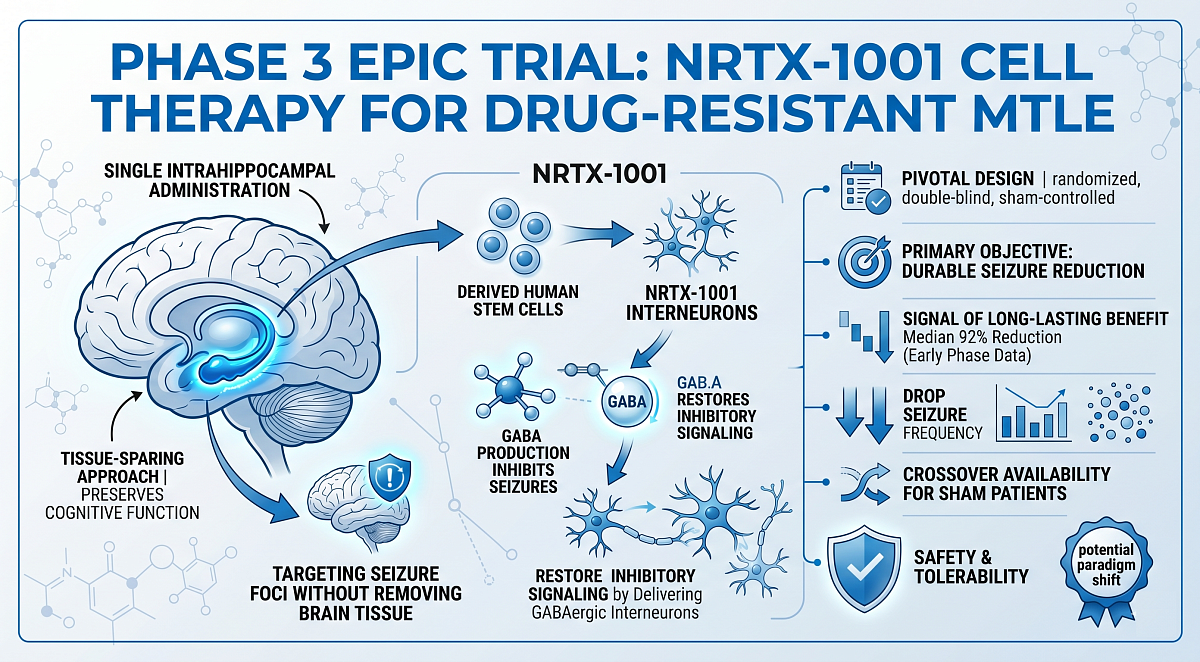
A pivotal 2:1 randomized, sham-controlled design tests whether intrahippocampal NRTX-1001 reduces 28-day average seizure frequency at 4–6 months versus sham after a 10-week baseline. Secondary/exploratory assessments include responder rates, EEG effects, neurocognition, mood, quality of life, and safety, with crossover availability for sham patients and immunosuppression taper at 1 year. Phase 3 EPIC trial design highlights NRTX-1001 cell therapy as a potential single-administration approach to achieving durable seizure reduction in drug-resistant MTLE.
Details of the phase 3 EPIC trial evaluating NRTX-1001 (Neurona), an investigational GABAergic interneuron cell therapy, highlight a potential tissue-sparing approach to reducing seizure frequency in adults with drug-resistant mesial temporal lobe epilepsy (MTLE), according to an abstract from the 2026 American Academy of Neurology (AAN) Annual Meeting.
The study, attributed to Manher Joshi, MD, chief medical officer at Neurona, and colleagues, outlines a multicenter, randomized, double-blind, sham-controlled trial designed to serve as a pivotal evaluation of the therapy. Adults with drug-resistant MTLE will undergo a 10-week baseline period before randomization (2:1) to receive a single intrahippocampal administration of NRTX-1001 or a sham procedure, with both groups receiving protocol-defined immunosuppression.
What is NRTX-1001?
NRTX-1001 is made from human stem cells that have been developed into interneurons (like the cells already in the brain). NRTX-1001 cells produce GABA, a neurotransmitter in the brain thought to inhibit seizure activity. NRTX-1001 is a first-in-class regenerative cell therapy derived from human pluripotent stem cells, designed to restore inhibitory signaling by delivering medial ganglionic eminence–like GABAergic interneurons directly into seizure foci.
Unlike resective or ablative procedures, the approach aims to rebalance neural circuitry without removing brain tissue, an important consideration for patients at risk of cognitive decline with conventional surgery.
Building on Phase 1/2 Success and Preclinical Findings
The phase 3 program builds on a growing body of early-phase and translational data. In an ongoing phase 1/2 study of adults with unilateral MTLE, NRTX-1001 has been well tolerated, with no treatment-related serious adverse events reported and signals of durable seizure reduction. Company-reported data presented in 2025 demonstrated a median 92% reduction in disabling seizures in the low-dose cohort, with 80% of treated patients achieving greater than 80% seizure reduction during the primary evaluation period. Notably, among patients followed for up to 24 months, some maintained greater than 97% seizure reduction after a single administration, suggesting the potential for long-lasting benefit.
Preclinical findings further supported the biologic rationale for this approach. In chronic MTLE mouse models, a single intrahippocampal dose of NRTX-1001 led to sustained suppression of focal seizures, with transplanted interneurons demonstrating long-term survival, functional integration, and synaptic activity within host circuits. These effects were accompanied by reduced hippocampal pathology and improved survival, without evidence of adverse behavioral effects.
Addressing a Crucial Unmet Need
Drug-resistant MTLE remains one of the most challenging epilepsy subtypes, with many patients continuing to experience frequent seizures despite antiseizure medications and facing limited options beyond invasive surgical interventions. If successful, the EPIC trial may help define whether a single-administration, cell-based therapy can provide durable seizure control while preserving cognitive function, representing a potential shift in the treatment paradigm for focal epilepsy.