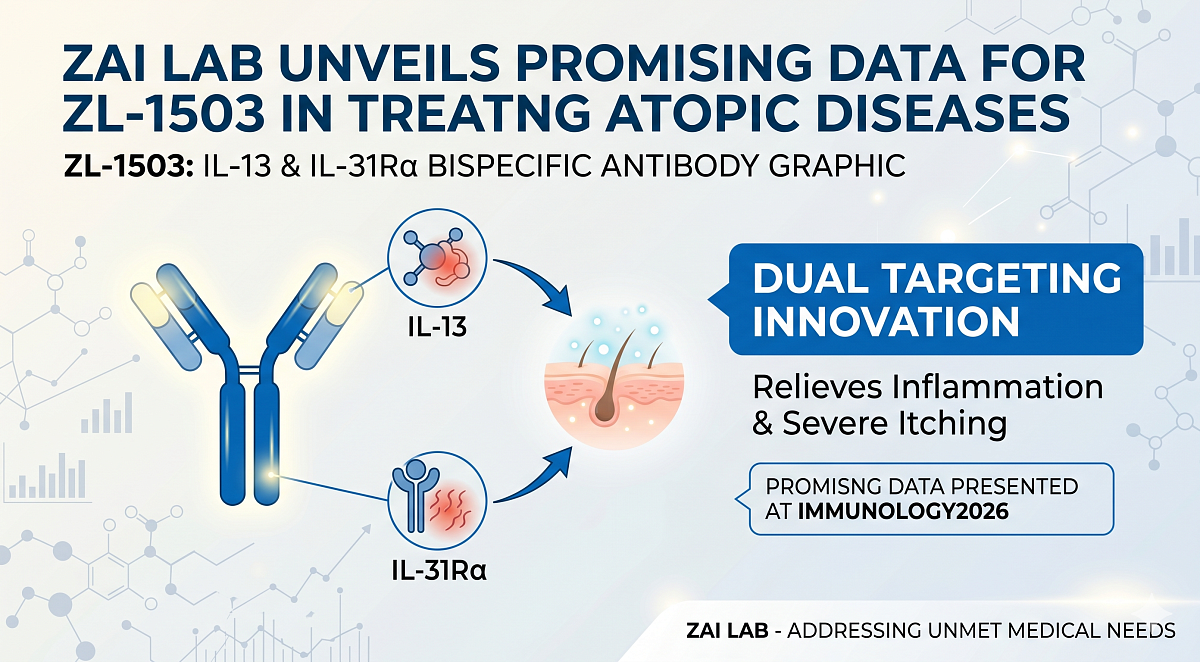
Zai Lab Limited shared new insights from a preclinical study on ZL-1503, an innovative bispecific antibody targeting IL-13 and IL-31Ralpha. The results indicate that this therapy could effectively reduce severe itching and inflammation associated with atopic diseases. These findings were presented at the IMMUNOLOGY2026 conference held in Boston, MA, highlighting ZL-1503's potential as a groundbreaking treatment for moderate to severe atopic dermatitis and other conditions driven by IL-13 and IL-31.
The announcement from Zai Lab regarding ZL-1503 is significant as it addresses a critical area of unmet medical need in the treatment of atopic diseases, particularly atopic dermatitis. The preclinical study results suggest that a single administration of ZL-1503 can lead to sustained relief from symptoms for up to 112 days, showcasing its potential efficacy across various disease models. This dual-targeting mechanism could represent a breakthrough in therapeutic options for patients suffering from these conditions, which are often challenging to manage with existing treatments.
Zai Lab Ltd operates in the biotechnology sector, focusing on discovering, developing, and commercializing proprietary therapeutics that address unmet medical needs, particularly in oncology, autoimmune, and infectious diseases. With a market capitalization of approximately $2.63 billion, Zai Lab has a diverse pipeline of potentially first-in-class product candidates developed through in-house research and development and global partnerships. The company has successfully commercialized several products in China, including Zejula, Optune, Qinlock, and Nuzyra, and is poised for further expansion.
About ZL-1503
ZL-1503 is a potential first-in-class bispecific antibody targeting IL-13 and IL-31Rα engineered by Zai Lab’s in-house discovery engine to simultaneously suppress both inflammatory and pruritogenic (itch-causing) pathways implicated in AD and other IL-13 and IL-31-driven diseases, potentially offering faster onset of action and superior efficacy compared to single-pathway inhibition. ZL-1503 is currently being evaluated in the ongoing global Phase 1/1b clinical trial evaluating its safety, tolerability, pharmacokinetics, and efficacy. This clinical advancement is supported by preclinical studies in which ZL-1503 demonstrated the ability to simultaneously suppress inflammatory and pruritogenic pathways implicated in AD.
About Zai Lab
Zai Lab Limited is an innovative, research-based, commercial-stage biopharmaceutical company based in China and the United States. We are focused on discovering, developing, and commercializing innovative products that address medical conditions with significant unmet needs in the areas of oncology, immunology, neuroscience, and infectious disease. Our goal is to leverage our competencies and resources to positively impact human health.