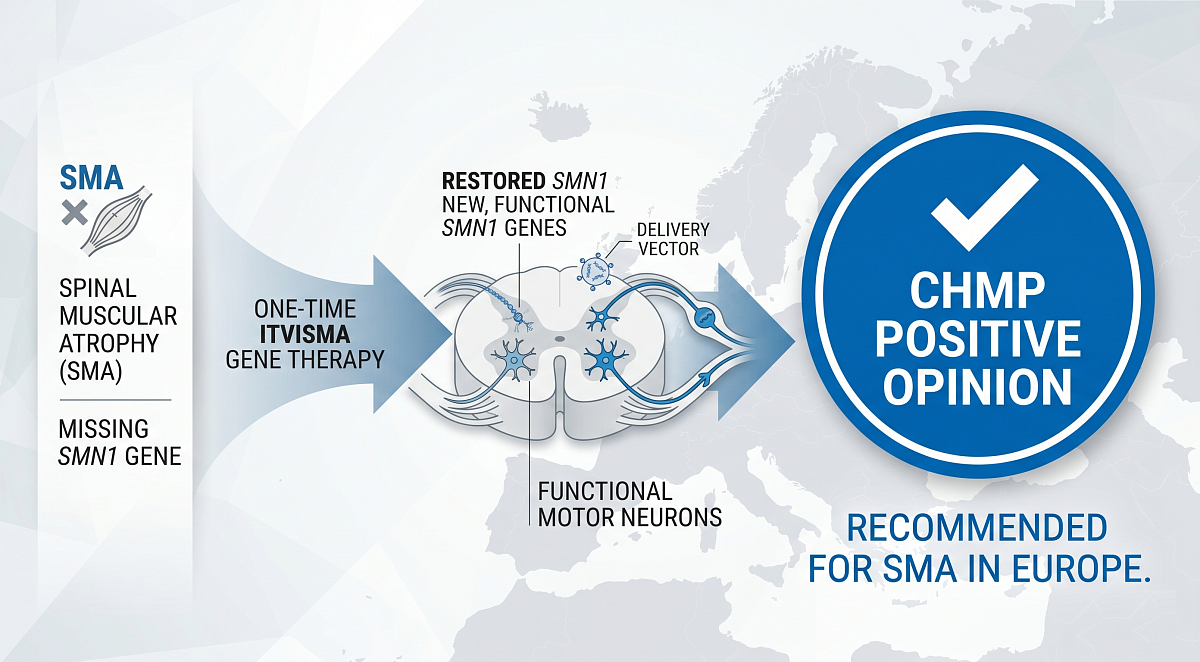
Meanwhile, five months after the FDA signed off on Novartis’ gene therapy Itvisma (onasemnogene abeparvovec), the CHMP has followed suit with a recommendation for the spinal muscular atrophy (SMA) treatment to be approved in Europe. Itvisma is the Swiss company’s SMA follow-on to Zolgensma, which in 2019 became the first gene therapy approved to treat a neuromuscular disease and gained attention for its $2.1 million price tag. In the U.S., Itvisma has been priced at $2.59 million, which is a significant reduction in the cost of standard treatments for SMA over a span of 10 years. After CHMP nod, Moderna CEO applauds EU's 'rigorous scientific review'.
While their active drug substances are identical, Itvisma is given directly to the central nervous system by way of an intrathecal injection near the spinal cord, whereas Zolgensma is administered intravenously. Because of that formulation, Itvisma can be given to heavier patients in a more concentrated dose.
About SMA
Spinal muscular atrophy (SMA) is a rare, genetic neuromuscular disease caused by a mutated or missing SMN1 gene. The SMN1 gene is responsible for producing most of the SMN protein a body needs for muscle function, including breathing, swallowing and basic movement. Without it, motor neurons are irreversibly lost, leading to progressive, debilitating muscle weakness. A second gene, the SMN2 gene, produces a small fraction (~10%) of functional SMN protein compared with the SMN1 gene. Individuals with more copies of the SMN2 gene generally have a less severe form of SMA than those with fewer copies. SMA has an estimated global prevalence of around 1 to 2 per 100,000 people, with an incidence of roughly 1 in 10,000 live births.
About Itvisma® (onasemnogene abeparvovec)
Itvisma, an adeno-associated virus 9 (AAV9)-based gene therapy, is uniquely designed to address the genetic root cause of SMA by providing a functional copy of the human SMN1 gene to improve motor function through sustained SMN protein expression with a single, one-time intrathecal injection. The brand name Itvisma has been conditionally accepted by EMA for the investigational product OAV101B (onasemnogene abeparvovec), but the product itself has not yet received marketing authorization from the EC. In the US, OAV101B has been approved under the brand name Itvisma.
Novartis has several key licenses supporting this program:
- An exclusive, worldwide license with Nationwide Children's Hospital to both the intravenous and intrathecal delivery of AAV9 gene replacement therapy for the treatment of all types of SMA.
- An exclusive, worldwide license from REGENXBIO for any recombinant AAV vector in its intellectual property portfolio for the in vivo gene replacement therapy treatment of SMA in humans.
- An exclusive, worldwide licensing agreement with Genethon for in vivo delivery of AAV9 vector into the central nervous system for the treatment of SMA.