Drug discovery services. Preclinical services
Leave your request for services (Custom chemistry, HTS, CADD, Integrated Drug Discovery, ADME, PK)
Leave your request for services (Custom chemistry, HTS, CADD, Integrated Drug Discovery, ADME, PK)
Integrated Drug Discovery
ChemDiv is dedicated to partnering in discovery and development of breakthrough therapies based on its unique chem-bio platforms: molecular glues, stabilizers and degraders; covalent and non-covalent binders; bifunctional molecules for synergistic effect and synthetic lethality; new generation ADCs; RNA and DNA editing.
Over the past 32 years ChemDiv has successfully completed hundreds of leads, candidates and new drug programs to treat cancer, neurological, infectious, cardiometabolic and immune diseases with pharma, biotech and academic partners around the globe. We are revolutionizing the field of drug discovery by harnessing the power of Hybrid Artificial Intelligence (AI) /Machine Learning (ML) which is curated by our scientists at each step in the process.
Our proprietary discovery platforms and advanced technologies are reducing timelines and efficiently using capital to deliver validated clinical assets in as little as 18 months. Let’s discuss how we can do this for you.
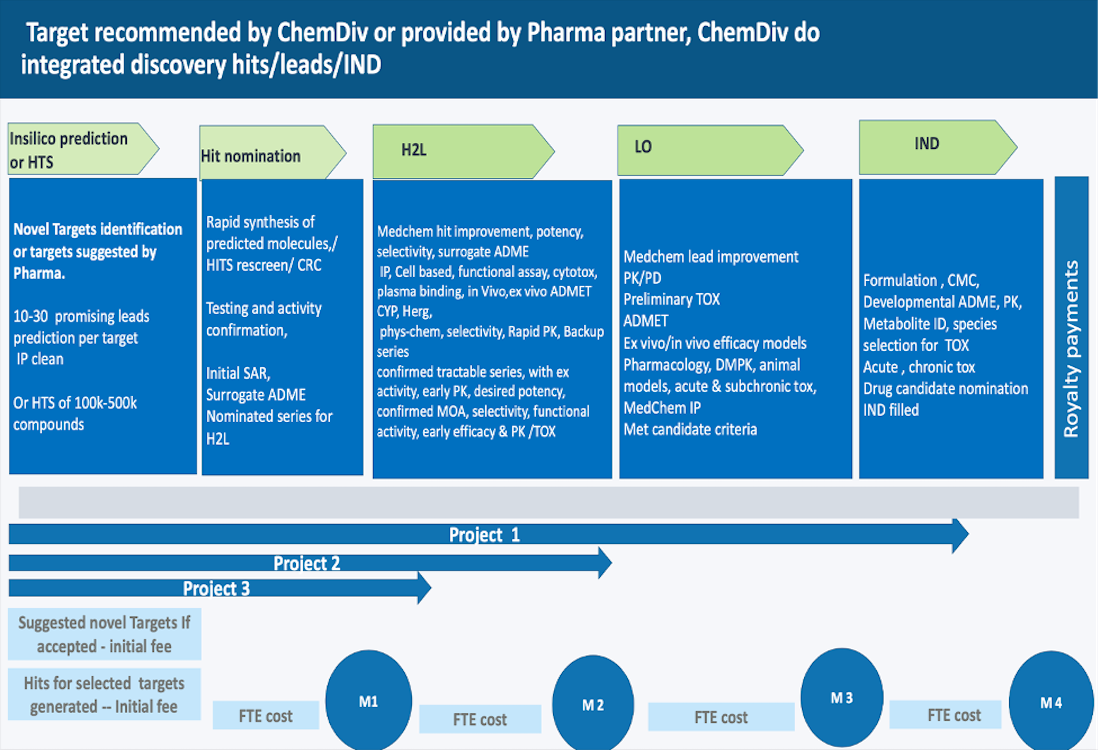
For every custom project, we assign an expert team of chemists and biologists to help our partners to define their research path and goals, including scientific and business plan.
We help you benefit from the value of our integrated drug discovery services that offer access to ChemDiv’s flexible organization as a one-stop and cost-effective solution.
Your project is in safe hands. Our teams’ intellectual contribution is highly valued by our customers and recognised by co-authorship in numerous published patents assigned to and held by our customer companies
Over the past 29 years ChemDiv has delivered hundreds of leads, drug candidates and new drugs in major therapeutic areas. The experience of ChemDiv’s team allows us to anticipate the requirements of modern preclinical research as well as develop new ideas for your research project. Our clients benefit from our expertise in consultancy together with our flexibility and scientific excellence to test their compounds. ChemDiv offers a wide range of Integrated Drug Discovery Services: lead optimization, high throughput screening, ADME services, DMPK services, etc. From in vitro assays to in vivo studies, we have the expertise and resources to help you every step of the way. Are you looking for a reliable partner to help you with preclinical service? Look no further than ChemDiv, a recognized CRO in drug discovery solutions.
Our Biology FTE and Chemistry FTE offers maximum flexibility and consistent access to our world-class team of experts throughout your drug development program in the fields of biology and medicinal chemistry.
Ready to take your drug discovery project to the next level? Contact chemdiv@chemdiv.com today
Successful Collaborations

Advantages of Integrated drug discovery services of ChemDiv
Integrated drug discovery approach offers a speed and efficient process towards challenging research tasks that each life sciences organization of any size is faced with. This outsourcing model presents one project team working through all phases in the discovery and development process and overlooking the transition of molecules from early hits through optimized leads into clinical candidates
Working with ChemDiv as Integrated drug discovery CRO is advantageous as it allows customers to observe competitive cost efficiency, high workflow transparency, simplified project management and problem solving, better anticipation of the future development needs and understanding project prospective.




























